|
T hat's the last potato I'll ever dig!" That statement was attributed to young Ernest Rutherford, a native of New Zealand.  
It releases radiation and changes into a new element. A radioactive element is one that breaks apart spontaneously. Some years later, the reason for that connection became obvious. Ramsay did not know why helium occurred in an ore of uranium. Swedish chemists Per Teodor Cleve (1840-1905) and Nils Abraham Langlet also discovered helium at about the same time in a mineral called cleveite. Credit for the earthly discovery of helium is sometimes given to two other scientists also. Then, in 1895, the English physicist Sir William Ramsay (1852-1916) found helium in a mineral of the element uranium. Was there an element on the Sun that did not exist on Earth? Had he made a mistake? Some scientists even made fun of Janssen.įor the next thirty years, chemists looked for helium on Earth. The name comes from the Greek word helios for " sun."įrench astronomer Pierre Janssen discovered helium after studying the Sun during a full solar eclipse.Ĭhemists did not know what to make of Janssen's discovery. The name helium was later suggested for this element. He concluded that there must be an element on the Sun that had never been seen on Earth. As he looked at the spectral lines, he was surprised to see some lines that could not be traced to any known element. Janssen examined light from the Sun with a spectroscope. Solar eclipses provide scientists with an unusual chance to study the Sun. All that remains is a thin outer circle (corona) of sunlight around the dark Moon. The Moon blocks nearly all of the Sun's light. A solar eclipse occurs when the Moon comes between the Sun and the Earth. In 1868, Janssen visited India in order to observe a full eclipse of the Sun. This principle led to the discovery of helium. They can identify an element by recognizing its distinctive spectral lines even when they can't actually see the element itself. The spectroscope gives scientists a new way of studying elements. Every element has its own distinctive spectrum. These lines are called the element's spectrum. In the case of sodium, the yellow light is broken into a series of yellow lines. These basic parts consist of a series of colored lines. The flame looks quite different, however, when viewed through a spectroscope.Ī spectroscope contains a triangular piece of glass (called a prism) that breaks light into its basic parts. For example, a lump of sodium metal will burn with a yellow flame. The spectroscope is a device for studying the light produced by a heated object. One of the most powerful instruments for studying chemical elements is the spectroscope. Welding operations, in lead detection systems, in neon signs, and to protect objects from reacting with oxygen. It is used in low-temperature research, for filling balloons and dirigibles (blimps), to pressurize rocket fuels, in Richardson (1937-).įor an inactive gas, helium has a surprising number of applications. The Nobel Prize in physics for 1996 was awarded to three Americans who discovered superfluidity. It can also squeeze through very small holes that should be able to keep it out. It can flow upwards out of a container, against the force of gravity. For example, at very low temperatures it can become superfluid.Ī superfluid material behaves very strangely. Helium has some interesting and unusual physical properties. He found proof that a new element existed in the Sun. In 1868 French astronomer Pierre Janssen (1824-1907) studied light from the Sun during a solar eclipse. Helium was first discovered not on Earth, but in the Sun. The hydrogen atom has one proton, one electron, and no neutrons. 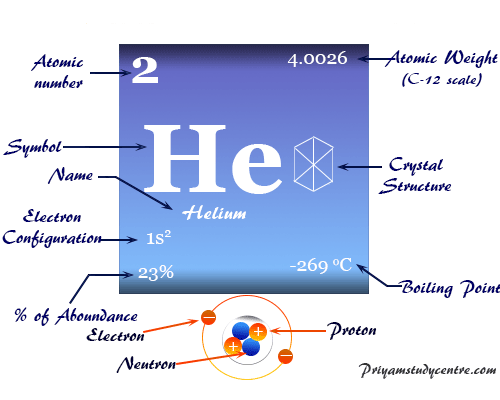
Only the hydrogen atom is simpler than a helium atom. Its atoms consist of two protons, two neutrons, and two electrons. Helium is also the second simplest of the chemical elements. Only hydrogen occurs more often than helium. Helium is the second most abundant element in the universe. In fact, no compounds of helium have ever been made. It will not combine with other elements or compounds. Inert means that an element is not very active. The noble gases are also called the inert gases. The periodic table is a chart that shows how the elements are related to one another. 
The noble gases are the elements in Group 18 (VIIIA) of the periodic table. Helium is a member of the noble gas family. Note: This article, originally published in 1998, was updated in 2006 for the eBook edition.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
